Rafie Hamidpour1,2* and Luay Rashan2
1Department of Herbal Medicine, Pars Bioscience Research Center, Leawood, Kansas, USA
2Biodiversity Center, Medicinal Plants Division, University of Dhofar, Oman
*Corresponding Author:
Dr. Rafie Hamidpour
Pars Bioscience Research Center USA
14109 Cambridge Lane, Leawood
Kansas 66224, USA
Tel: (913) 432-0107
Fax: (913) 432-5708
E-mail: rafie@parsbioscience.com
Received date: August 03, 2017; Accepted date: August 26, 2017; Published date: August 31, 2017
Citation: Hamidpour R, Rashan L (2017) A Novel Remedy for Irritable Bowel Syndrome. Transl Biomed. Vol.8 No.3:125. doi:10.21767/2172-0479.100125
Commentary
Irritable bowel syndrome (IBS) is a common disorder which affects the digestive tract and the large intestine in particular. Common symptoms of IBS include cramping, abdominal pain, bloating, gas, diarrhea, and constipation. These complications are often long-lasting and therefore require extensive treatment.
Symptoms of irritable bowel syndrome are often uncomfortable and sometimes even debilitating, but IBS does not cause changes in bowel tissue or increase the risk of colorectal cancer. Only a small group of people with irritable bowel syndrome has severe signs and symptoms. Some patients can control their symptoms by managing diet, lifestyle, and stress, while others need more intensive treatment and medications [1]. Because many of these medications have potentially harmful side effects, Pars Bioscience is pleased to introduce EDS2, a novel herbal remedy designed to help patients overcome irritable bowel syndrome (Figure 1).
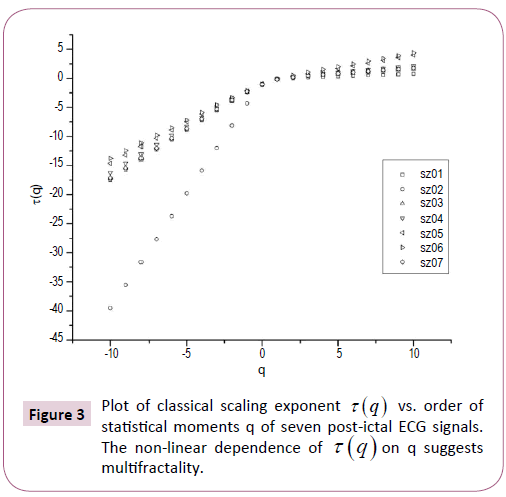
Figure 1: Pars Bioscience, LLC.
EDS2 is a remedy based on food ingredients that exhibit an action on the bowel. This OTC product contains a blend of complementary herbs to help slow digestion, reduce intestinal gas bloating, and alleviate stomach pain after eating. It is particularly designed for those suffering from irritable bowel syndrome [2-5].
Properties of EDS2
1. Offers a safe and quick relief of digestive complaints such as colic, indigestion,
2. Aids the digestive system and gastrointestinal tract.
3. Composed of potent carminative natural herbs.
4. Alleviates indigestion.
5. Enhances digestive enzyme release.
6. Helps relieve many symptoms of heartburn.
7. Relieve digestive disturbances including bloating and flatulence.
8. Helps in restoring normal stool weight and transit time in patients who are constipated.
9. Offers significant improvements in both IBS symptoms and the inflammatory profile.
Uniqueness and safety of EDS2
1. Consists of powerful blend of three well-researched and clinically-substantiated herbs.
2. Safely taken with no known side effects.
3. This product contains all-natural ingredients.
How EDS2 works
1. Exerts effect on the colon resulting in significant improvement (70% to 80%) in connection with the abdominal colic, good relief (60?70%) from flatulence and indigestion, and significant improvements (65% to 75%) from GIT disturbances and belching [2-6].
2. Restores the peristalsis movement.
3. Suppresses bloating, indigestion, belching and heartburn [7,8].
4. Effectively breaks down food to nourish the body, increases nutrient digestion, and maximizes absorption.
5. Helps ease stomach and intestinal discomfort and relieves acid digestion [8].
6. Showed significant reduction in the circulating levels of pro-inflammatory cytokines including IL-6, IL-7 and others 3.
EDS2 was developed following a careful preclinical study both in vitro and in vivo for more than two years. On the other hand, it was tested on more than 3000 individuals suffering from flatulence with gases, abdominal colic with constipation alternated with diarrhea, gastro-intestinal disturbances (GIT), indigestion and belching. More than 70% of these cases had received different therapies including antispasmodics, serotonin agonists and other anti-cholinergic treatments.
These studies showed than more than 80% of the individuals used EDS2 showed positive results especially those IBS –patients with constipations. During the period of these studies it was observed that EDS2 exerted effects on the colon resulted into significant improvements (70%-80%) in connection with the abdominal colic, good relieve (60%-70%) from GIT-disturbances and belching It also restored the peristaltic movement of the colon and showed significant improvements for bloating, indigestion and heartburn and it was also effective in breaking down food to nourish the body, increased nutrient digestion and maximized absorption.
Note: Can also be supplied as powder or as sachet form (Figure 1).
References
- Mayo Clinic (2014) Irritable bowel syndrome. Mayo Foundation for Medical Education and Research, USA.
- Dubinin AV, Kabanov AV, Kirkin BV, Kolkunova GK, Igorianova NA (1987) Bran in the treatment of irritable bowel syndrome. Vopr Pitan 1: 13-16.
- Sofi F, Whittaker A, Gori AM, Surrenti E, Abbate R, et al. (2014) Effect of triticum turgidum Subsp. Turanicum wheat on irritable bowel syndrome. A double-blinded randomized dietary intervention trial. Br J Nutr 111: 1992-1999.
- Ijaz H, Tulain UR, Qureshi J, Danish Z, Musayab S, et al. (2017) Nigella sativa (Prophetic Medicine): A Review. Pak J Pharm Sci 30: 229-234.
- Gholamnezhad Z, Havakhah S, Boskababy NH (2016) Preclinical and clinical effects of Nigella sativa and its constituent, thymoquinone: A review. J Ethnopharmacol 190: 372-386.
- Muller-Lissner SA (1988) Effects of wheat bran on weight of stool and gastrointestinal transit time: A meta-analysis. Br Med J (Clin Res Ed) 296: 615-617.
- Gargam C, Khan SA, Ansari SH, Suman A, Garg M (2009) Chemical composition, therapeutic potential and perspectives of Foeniculum vulgare. Pharmacognosy Review 3: 346-352.
- Health Canada Sweet Fennel Monograph: EMEA (2007) European Medicines Agency. Community Monograph on FV. Miller. London (UK); EMEA Committee on Herbal Medicinal Products (HMPC).
20556
References
- Mayo Clinic (2014) Irritable bowel syndrome. Mayo Foundation for Medical Education and Research, USA.
- Dubinin AV, Kabanov AV, Kirkin BV, Kolkunova GK, Igorianova NA (1987) Bran in the treatment of irritable bowel syndrome. Vopr Pitan 1: 13-16.
- Sofi F, Whittaker A, Gori AM, Surrenti E, Abbate R, et al. (2014) Effect of triticum turgidum Subsp. Turanicum wheat on irritable bowel syndrome. A double-blinded randomized dietary intervention trial. Br J Nutr 111: 1992-1999.
- Ijaz H, Tulain UR, Qureshi J, Danish Z, Musayab S, et al. (2017) Nigella sativa (Prophetic Medicine): A Review. Pak J Pharm Sci 30: 229-234.
- Gholamnezhad Z, Havakhah S, Boskababy NH (2016) Preclinical and clinical effects of Nigella sativa and its constituent, thymoquinone: A review. J Ethnopharmacol 190: 372-386.
- Muller-Lissner SA (1988) Effects of wheat bran on weight of stool and gastrointestinal transit time: A meta-analysis. Br Med J (Clin Res Ed) 296: 615-617.
- Gargam C, Khan SA, Ansari SH, Suman A, Garg M (2009) Chemical composition, therapeutic potential and perspectives of Foeniculum vulgare. Pharmacognosy Review 3: 346-352.
- Health Canada Sweet Fennel Monograph: EMEA (2007) European Medicines Agency. Community Monograph on FV. Miller. London (UK); EMEA Committee on Herbal Medicinal Products (HMPC).






