Keywords
Yarrowia lipolytica; Alkaline extracellular protease; Marine yeast; Enzyme
Introduction
Along the vast biodiversity of animals, plant and seaweeds; marine environments contain innumerable microorganisms including microalgae and yeast each; with distinctive metabolic functions capable of producing bio-actives substances. The marine yeasts; initially discovered and isolated from the Atlantic Ocean (van Uden and Castelo-Branco, 1963); were subsequently isolated from various sources including seawater, seaweeds, fish, and marine sediments (Kutty and Philip, 2008). Beside its nutritional quality and its possible utilization as animal or aquaculture feed, marine yeast represent a substantial and a competitive source of enzymes. The global market for industrial enzymes is now well established reaching around $5.0 billion in 2016 with a potential for growth rate of 4.7% until 2021 (Dewan, 2017). With several distinctive and promising attributes (Zaky et al., 2014), marine yeasts produce various enzyme such as inulinase, amylase, cellulase, exo β-1,3-glucanase, and protease (Liu et al., 2008; Zhang et al., 2009; Ni et al., 2009; Peng et al., 2011; Xu et al., 2012; Zhang et al., 2015; Rong et al., 2015 and Chi et al., 2016).
Proteases are considered among the most important groups of industrially and commercially produced enzyme with application in various sectors such as biotechnology, food, pharmaceutical process, cosmetics, and detergent (Anwar et al., 1998; Bagewadi et al., 2011; Vermelho et al., 2012; Alnahdi., 2012; Kasana et al., 2011; Pokora et al., 2017).
Yarrowia lipolytica is one of the most extensively studied ‘‘non-conventional’’ yeasts which is currently used as a model for the study of protein secretion, peroxisome biogenesis, dimorphism, degradation of hydrophobic substrates, and several emerging fields (Fickers et al., 2005). According to Singhal (2012) and Ogrydziak (2013), all Yarrowia lipolytica strains can produce acid and alkaline extracellular protease with a high rate of alkaline extracellular (1-2% of total cell protein) (Motabe et al., 1988; Madzak et al., 2004). In a previous study (Bessadok et al., 2015), proteases secreted by the strain of Yarrowia lipolytica YlTun15, was extracted and its alkaline nature was determined. The aim of the current study is to characterize the alkaline extracellular protease.
Materials and Methods
Determination of the protease activity
Yeast Yarrowia lipolytica YlTun15 cell culture was centrifuged at 8000 rpm/4°C/30 min and the resultant supernatants were concentrated (Amicon Bioseparation, Merck). Thereby, 0.5 ml of the supernatant was mixed with 1 ml of 0.5% of casein solution in glycine-NaOH buffer (0.05M, pH 9.0). The mixture was incubated at 45°C for 30 min and 2 ml of trichloroacetic acid (10%) was added to the mixture immediately to stop the reaction. The reaction mixture was centrifuged at 10000 rpm/4°C (Ma et al., 2007 and Chi et al., 2007). Tyrosine content was determined calorimetrically at 650 nm according to Lowry method (1951). The enzyme activity was defined as the amount of the enzyme that liberated 1 μg of tyrosine per minute. Protein concentration was measured by the method of Bradford (Bradford, 1976).
Effect of pH and temperature on enzyme activity and the stability
The effect of pH on the purified alkaline protease activity was determined by incubating the purified enzyme at pH 6 to 10 using the standard assay conditions. The buffers used were 0.1 M sodium phosphate (pH 6.0 to 8.0), 0.1M glycine-NaOH (pH 9 to 10). The optimal temperature for the enzyme activity was determined at 15°C, 25°C, 45°C and 60°C in the same buffer as described in the first section. The pH stability was tested via 12 h pre-incubation of the crude enzyme in appropriate buffers that had the same ionic concentrations at different pH values ranging from 6 to 10 at 4°C. The activities of protease were measured immediately after this treatment with the standard method as mentioned previously. Temperature stability of the purified enzyme was tested by preincubating the enzyme at different temperatures (15°C, 25°C, 45°C and 60°C) for 1h, the relative activity was immediately measured (Ma et al., 2007).
SDS-PAGE electrophoresis
The profile of the protease was analysed in non-continuous denaturing Sodium Dodecyl Sulfate-Polyacrylamide gel electrophoresis SDS-PAGE (Laemmli, 1970) in a mini Protean II Electrophoresis System (Bio-Rad, Hercules, CA.). 5 μl of protein molecular weight marker of 250 KDa (Precision Plus Protein ™ Dual Xtra standard; Criterion 4-20%Tris-HCl; Bio-Rad) was added into gel. The gel was than stained using Coomasie Brilliant Blue (R-250) method ans analysed with Digi-Doc-IT System (UVP, Upland, Ca).
Effect of different metal ions on the enzyme activity
To examine the effects of the different metal ions on the protease activity, enzyme assay was made for 1 h in the reaction mixture as described previously with various metal ions at a final concentration of 1 mM to 5 mM. The activity assayed in the absence of metal ions was defined as the control. The metal ions tested included Zn2+, Cu2+, Mg2+, Ca2+, Mn2+, Fe2+, Na2+ K+, Hg+ and Ni+ (Ma et al., 2007).
Effect of protein inhibitors on the enzyme activity
The effects of inhibitors on protease activity were measured in the reaction mixture. Such inhibitors included ethylenediaminetetraacetic acid (EDTA), phenylmethylsulfonyl fluoride (PMSF) and sodium dodecylsulfate (SDS) at a final concentration ranging from 1 to 5 mM, respectively. The crude enzyme was pre-incubated with the respective compound for 30 min at 0°C, followed by the standard enzyme assay as described in the preceding text. The activity assayed in the absence of the protein inhibitors was defined as the control (Chi et al., 2007).
Effect of storage time on the enzyme stability
The storage stability of the enzyme at -20°C was analyzed as described by Beena et al. (2012). The enzyme was stored at the corresponding temperature and the residual activity was recorded by the standard assay procedure as described previously after exactly 4 months for a period of 18 months. The relative activity at day one was considered as 100%.
Statistical analysis
Data were subjected to analyses of variance at the 5% level using SPSS 24.0 software and the Tuckey-test was performed to separate differences among means.
Results and Discussions
Gel electrophoresis
The profile of the protein was determinate with SDS-PAGE. Figure 1 shows that the purified enzyme presented two bands but after a dilution (1:4), only one band persisted with the same intensity. Thus, the crude alkaline extracellular enzyme has a relative molecular weight about 35 kDa. According to Matoba and Ogrydziak (1989); Barth and Gaillardin (1997), Yarrowia lipolytica can produce an Alkaline extracellular protease with a molecular weight of 32 kDa which processed from a 55 kDa glycosylated precursor. It was also mentioned that the molecular weight of alkaline protease secreted by microorganism ranged between 15 to 45 kDa (Kumar and Takagi, 1999).
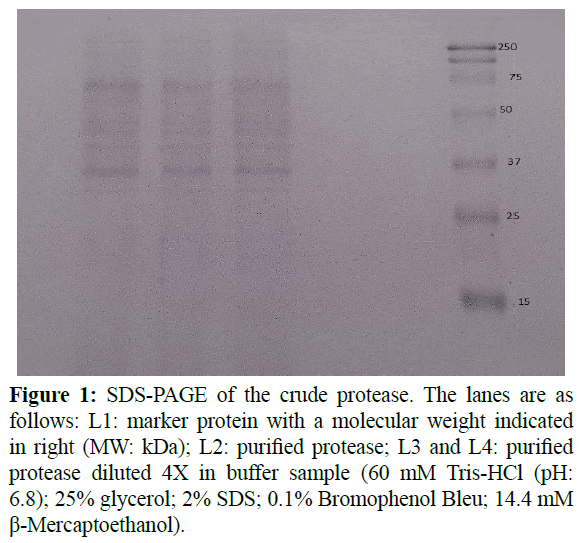
Figure 1: SDS-PAGE of the crude protease. The lanes are as follows: L1: marker protein with a molecular weight indicated in right (MW: kDa); L2: purified protease; L3 and L4: purified protease diluted 4X in buffer sample (60 mM Tris-HCl (pH: 6.8); 25% glycerol; 2% SDS; 0.1% Bromophenol Bleu; 14.4 mM β-Mercaptoethanol).
Effect of temperature on the stability and the optimum activity of the enzyme
In this study, the Yarrowia lipolytica alkaline protease activity measured at different temperatures ranging from 15°C to 60°C (Figure 2); revealed a maximum activity at 45°C. Similar result was reported for Aureobasidium pullulans (Chi et al., 2007; Ma et al., 2007), but a lower temperature (37°C) for optimal protease activity was found B. subtillus (Alam et al. 2017). The residual protease activities were 97.29 ± 0.11% and 95.85 ± 0.34% at 15 and 25°C respectively. From the temperature of 45°C, the relative activity drops significantly and the enzyme becomes almost inactive at 60°C in 30 minutes. Such result suggests that this enzyme is thermosensitive at temperature above 45°C but is stable at temperature ranging from 15-25°C.
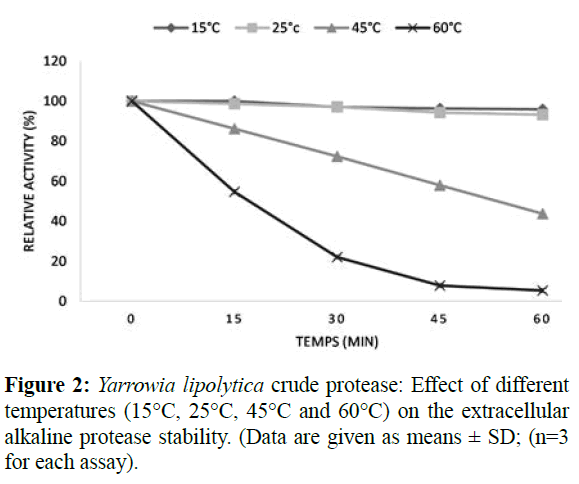
Figure 2: Yarrowia lipolytica crude protease: Effect of different temperatures (15°C, 25°C, 45°C and 60°C) on the extracellular alkaline protease stability. (Data are given as means ± SD; (n=3 for each assay).
Effect of pH on the stability and the optimum activity of the enzyme
It well established that pH is a key factor influencing the ionization state of the amino acid or ionization of the substrate and thereby the expression of the enzyme activity (Bezawada et al., 2011). In the present study, the relative protease activity measured at different pH ranging from 6 to 10 (Figure 3) showed maximum activities at pH 9 (88.69 ± 1.71). In this case, the enzyme stability was maintained over 12 h at 4°C. The average residual activity of the extracted protease showed a linear and significant decrease with pH drop. This result is consistent with that found for protease extracted from other micro-organisms (Kumar and Tagaki, 1999; Ma et al., 2007), but a higher pH value (11) was found for an optimal activity of protease from Aspergillus terreus (Niyonzima and More, 2015), which was also inactive at low pH. It was also reported that a large amount of alkaline protease could be secreted under neutral condition and three acids protease can be secreted by Y. lipolytica (Barth and Gaillardin, 1997). Moreover, Li et al. (2010) report that despite M. reukaufii W6b was isolated from alkaline marine environment (pH 8), it is completely unknown if this strain can produce this enzyme in natural sea sediment.
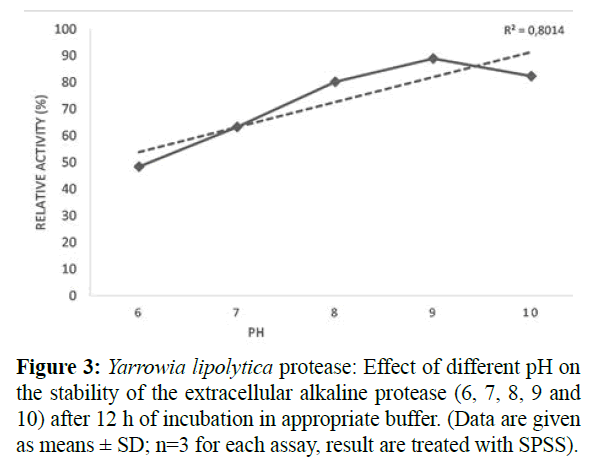
Figure 3: Yarrowia lipolytica protease: Effect of different pH on the stability of the extracellular alkaline protease (6, 7, 8, 9 and 10) after 12 h of incubation in appropriate buffer. (Data are given as means ± SD; n=3 for each assay, result are treated with SPSS).
Effect of metal ion on the enzyme activity
The result presented in Table 1 shows that K+, Na2+, Mg2+ and Ca2+ had no effect on the protease activity. Some of the metal ions such as Ca2+, Mn2+, and Mg2+ increased and stabilized protease activity; this may be due to the activation by the metal ions (Bezawada et al., 2011; Kumar et Takagi, 1999). However, Figure 4 illustrate that metal ions including Ni+, Zn2+ and Fe2+ showed maximum inhibition on enzyme activity up to 60%, while Hg+ inhibited the enzyme activity up to 35%. Analogous results were found for the A. pullulan, A. terreus and B. subtilis (Ma et al., 2007, Niyonzima and More, 2015, and Alam et al., 2017).The inhibitory effect of mercury was due to the fact that it reacts with protein thiol groups and with histidine and tryptophan residues (Bezawada et al., 2011, Barth and Gaillardin, 1997). The Cu2+ and Mn2+ cations seem to have an antagonistic effect on the enzyme activity (Figure 5).
| |
Control |
1 mM |
2 mM |
3 mM |
4 mM |
5 mM |
| K+ |
100.86 ± 1.92 |
101.08 ± 0.97 |
101.13 ± 0.99 |
98.94 ± 1.37 |
99.42 ± 0.69 |
98.8 ± 2.31 |
| Na2+ |
100.86 ± 1.92 |
98.66 ±1.66 |
101.23 ± 1.21 |
99.40 ± 0,82 |
98.8 ± 1.97 |
100 ± 0.11 |
| Mg2+ |
100.86 ± 1.92 |
101.26 ± 1.32 |
107.66 ± 1,36 |
98.66 ± 1.68 |
103.9 ± 1.97 |
109.85 ± 1.35 |
| Ca2+ |
100.86 ± 1.92 |
101.75 ± 2.04 |
100.61± 0.09 |
99.52 ± 2.39 |
100.61 ± 1.93 |
108.04 ± 3 |
Table 1: Yarrowia lipolytica protease: Effect of Cations on the relative activity of the crude extracellular.
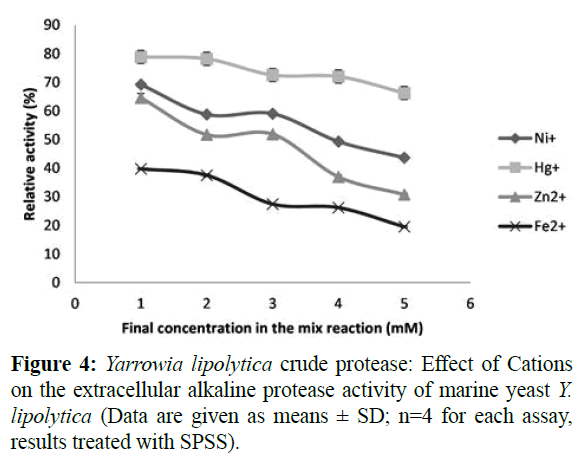
Figure 4: Yarrowia lipolytica crude protease: Effect of Cations on the extracellular alkaline protease activity of marine yeast Y. lipolytica (Data are given as means ± SD; n=4 for each assay, results treated with SPSS).
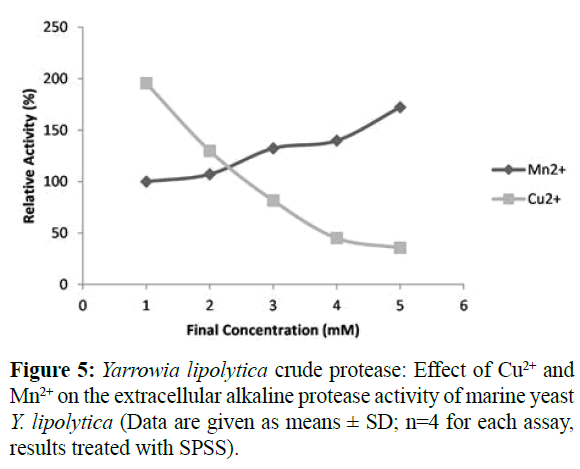
Figure 5: Yarrowia lipolytica crude protease: Effect of Cu2+ and Mn2+ on the extracellular alkaline protease activity of marine yeast Y. lipolytica (Data are given as means ± SD; n=4 for each assay, results treated with SPSS).
Effect of the protein inhibitors
Yeast alkaline protease activity reacted differently with the three tested inhibitors (Figure 6) with PMSF having the highest inhibitory effect at the lowest concentration of 2 mM in the broth. With a concentration of 5 mM PMSF in the broth, a drastic inhibition of the protease activity occurred (1.47%). Such results confirm that this extracellular enzyme was a serine protease (Castagliuolo et al., 1996; Moteba et al., 1997; Barth and Gaillardin, 1997; Alam et al., 2017) and it is belong to subtilism family. Up to 5 mM, the protease activities in the presence of EDTA and SDS were 74% and 55% respectively. Correspondingly, proteases from Aspergillus terreus and Aspergillus tamari exhibited a stability in the presence of EDTA (Anandan et al., 2007, Niyonzima and More, 2015). Such finding approve that Yarrowia lipolytica protease do not belong to the metalloprotein group, and could be of interest to the detergent industry as the stability of protease in the presence of chelating agents such as EDTA is a prerequisite for any detergent enzyme (Beg and Gupta, 2003).
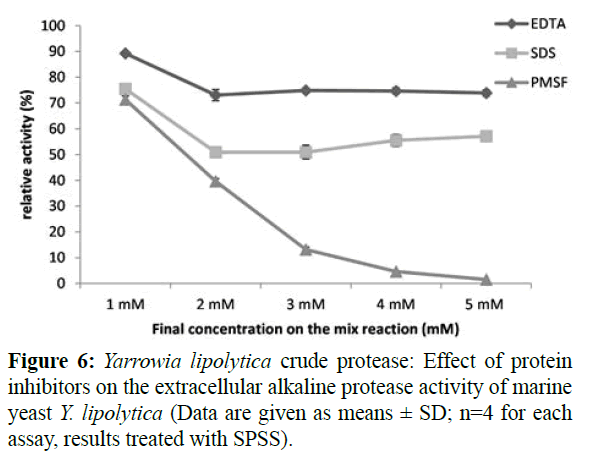
Figure 6: Yarrowia lipolytica crude protease: Effect of protein inhibitors on the extracellular alkaline protease activity of marine yeast Y. lipolytica (Data are given as means ± SD; n=4 for each assay, results treated with SPSS).
Effect of storage on the stability of the enzyme activity
Monitoring the effect of storage at -20°C for one year on protease activity showed a significant decrease in relative activity to reach 23.17 ± 2.11 in January 2017. This decrease might be explained by the ice crystal formation that inactivates enzymes (Beena et al., 2012). Comparing to this result, Niyonzima and More (2015) found that for Aspergillus terreus the protease decrease with 9% after 40 days storage in -20°C.
Conclusion
The purified extracellular alkaline protease extracted from Yarrowia lipolytica YlTun15 shows promising proprieties of stability and activities that may be used in profile various applications. Further, this study will be completed with proteomic analysis to sequence and well characterize this enzyme.
Acknowledgment
This work was conducted within the project entitled Biotechnologie Marine Vecteur d’Innovation et de Qualité - BIOVecQ PS1.3_08 co-financed by the European Union through the European Neighborhood and Partnership Instrument (ENPI) Italy-Tunisia program and the Tunisian Ministry of Higher Education.
20978
References
- Alam, M.G., Uddin, M.E., Rahman, S., Ahmed, T., Karim, M.R., et al. (2017) Protease activity of extracellular enzyme produced by B. subtilis isolated from soil. Int JEnvironAgricBiotechnol2, 382-388.
- Alnahdi, H.S.(2012) Isolation and screening of extracellular protease produced by new isolated Bacillus sp. J AppPharmaSci2,71-74.
- Anandan, D., Marmer, W.N., Dudley, R.L. (2007) Isolation, characterization and optimization of culture parameters for production of an alkaline protease isolated from Aspergillustamarii. J IndMicrobiolBiotechnol34,339–347.
- Anwar, A., Saleemuddin, M. (1998)Alkaline protease: a review. BioresourTechnl64, 175–183.
- Bagewadi, Z.K., Garg, S.D., Deshnur, P.B., Shetti, N.S., Banne, A.A. (2011) Production dynamics of extracellular alkaline protease from Neisseria sps isolated from soil.BiotechnolBioinfBioeng1, 483–493.
- Barth, G.,Gaillardin, C. (1997) Physiology and genetics of the dimporphic fungus Yarrowialipolytica. FEMS Microbial Rev 19, 219-237.
- Beena, A.K., Geevarghese, P.I., Jayavardanan, K.K. (2012) Detergent potential of a spoilage protease enzyme liberated by a psychotrophic spore former isolated from sterilizedskim milk. Am J Food Technol7,89-95.
- Beg, Q.K., Gupta, R. (2003) Purification and characterization of an oxidation-stable, thiol-dependent serine alkaline protease from Bacillus mojavensis. Enzyme MicrobiolTechnol32,294–304.
- Bessadok, B., Masri, M., Breuck, T.,Sadok, S. (2015) Isolation and screening for protease activity by marine microrganisms. Bull. Ins. Natn. Scien. Tech.Mer de Salammbô, vol. 42, 21-23.
- Bezawada, J., Yan, S., John, R.P., Tyagi, R.D., Surampalli, R.Y. (2011) Recovery of Bacillus licheniformis Alkaline Protease from Supernatant of Fermented Wastewater Sludge Using Ultrafiltration and Its Characterization. Biotechnology Research International p:11.
- Bradford, M.M. (1976) A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem72, 248–253.
- Castagliuolo, I., LaMont, J.T., Nikulasson, S.T.,Pothoulkis, C. (1996)Saccharomyces boulardii protease inhibits Clostridium difficile toxin a effects in the rat Ileum. Infection and Immunity 64,5225-5232
- Chi, Z., Liu, G., Lu, Y., Jiang, H., Chi, Z.M. (2016) Bio-products produced by marine yeasts and their potential applications.BioresourceTechnol202,244-252.
- Chi, Z., Ma, C., Wang, P., Li, H.F. (2007) Optimization of medium and cultivation conditions for alkaline protease production by the marine yeast Aureobasidiumpullulans.BioresourceTechnol98,534-538.
- Dewan, S.S. (2017) Global markets for enzymes in industrial applications. BCC Research, BIO030J.
- Fickers, P., Benetti, P.H., Wache, Y., Marty, A., Mauersberger, S., et al. (2005) Hydrophobic substrate utilisation by the yeast Yarrowialipolytica, and its potential applications. FEMS Yeast Research 5: 527–543.
- Kasana, R.C., Salwan, R., Yadav, S.K. (2011) Microbial Proteases: detection, production and genetic improvement. Crit Rev Microbiol37,262-276.
- Kumar, C.G., Takagi, H. (1999) Microbial alkaline proteases: From a bioindustrial viewpoint. BiotechnolAdv17, 561-594.
- Kutty, S.N., Philip,R. (2008) Review Marine yeasts -a review. Yeast 25, 465-483.
- Laemmli, U.K. (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature227, 680-685.
- Li, J., Peng, Y., Wang, X., Chi, Z. (2010) Optimum production and characterization of an acid protease from marine yeast Metschnikowiareukaufii W6b. J Ocean Univ China 9, 359-364.
- Liu, Z.Q., Li, X.Y., Chi, Z.M. (2008) Cloning, characterization and expression of the extracellular lipase gene from Aureobasidiumpullulans HN2-3 isolated from sea saltern.Antonie vanLeeuwenhoek 94, 245-255.
- Lowry, O.H., Rosebrougt, N.J., Farr, A.L., Randell, R.J. (1951) Protein measurement with folin phenol Rea. J BiolChem193,267-275.
- Ma, C., Ni, X., Chi, Z., Ma, L., Gao, L. (2007)Purification and Characterization of an Alkaline Protease from the Marine yeast Aureobasidiumpullulans for bioactive peptide production from different sources. Marine Biotech9,343-351.
- Madzak, C., Gaillardin C., Beckerich, J.M. (2004) Heterologous protein expression and secretion in the non-conventional yeast Yarrowialipolytica: a review. J Biotechnol109,63-81.
- Matoba, S., Morano, K.A., Klionsky, D.J., Kim, K.,Ogrydziakl, D.M. (1997)Dipeptidylaminopeptidase processing and biosynthesis of aIkaIineextraceIIular protease from Yarrowialipolytica.Microbiology 143, 3263-3272.
- Matoba, S., Ogrydziak, D.M. (1989)A novel location for dipeptidylaminopeptidase processing sites in the alkaline extracellular protease of Yarrowialipolytica. J BiolChem264, 6037-6043.
- Matoba, S., Fukayama, J., Wing, R.A., Ogrydziak, D.M. (1988) Intracellular precursors and secretion of alkaline extracellular protease of Yarrowialipolytica. Molecular and cellular Biology 8, 4904-4916.
- Ni, X.M., Yue, L.X., Chi, Z.M., Li, J., Wang, X.H., et al. (2009) Alkaline protease gene cloning from the marine yeast Aureobasidiumpullulans HN2-3 and the protease surface display on Yarrowialipolytica for bioactive peptide production. Mar Biotechnol11, 81-89.
- Niyonzima, F.N., More, S.S. (2015) Purification and characterization of detergent-compatible protease from Aspergillusterreus gr. 3 Biotech 5, 61–70.
- Ogrydziak, D.M. (2013) Acid and alkaline extracellular proteases of Yarrowialipolytica.Biotechnological Applications. Barth, G. (editor) p: 268.
- Peng, Y., Liu, G.L., Yu, X.J., Wang, X.H., Li, J., et al. (2011) Cloning of exo-b-1,3- glucanase gene from a marine yeast Williopsissaturnus and its overexpression in Yarrowialipolytica. MarBiotechnol13, 193–204.
- Pokora, M., Zambrowicz, A., Zabłocka, A., Dąbrowska, A., Szołtysik, M., et al. (2017)The use of serine protease from Yarrowialipolyticayeast in the production of biopeptides from denatured egg white proteins. Epub: No2016_1316.64.
- Rong, Y.J., Zhang, L., Chi, Z.M., Wang, X.H. (2015)Acarboxymethylcellulase from a marine yeast Aureobasidiumpullulans 98: Its purification, caractherization, gene cloning and Carboxymethylcellulase digestion. J Ocean Univ China 14, 913–921.
- Singhal, P., Nigam, V., Vidyarthi, A. (2012) Studies on production, characterization and applications of microbial alkaline proteases. International Journal of Advanced Biotechnology and Research 3,653-669.
- vanUden, N., Castelo-Branco, R. (1963) Distribution and population densities of yeast species in Pacific water, air, animals and kelp off southern California. LimnolOceanogr8, 323-329.
- Vermelho, A.B, Supuran, C.T, Guisan, J.M. (2012) Microbial enzyme: applications in industry and in bioremediation. Enz Res ID 980681.
- Xu, J.L., Zhang, X., Sun, H.Y., Chi, Z.M. (2012) Disruption of the gene encoding b-1,3- glucanase in marine-derived Williopsissaturnus WC91-2 enhances its killer toxin activity. Mar Biotechnol14, 261-269
- Zaky, A.S., Tucker, G.A., Daw, Z.Y.,Du, C. (2014) Marine yeast isolation and industrial application. FEMS Yeast Res 14, 813-825.
- Zhang, L.L., Liu, G.L., Chi, Z., Wang, G.Y., Wang, Z.P. et al. (2015) Cloning and characterization of an inulinase gene from the marine yeast Candida membranifaciens subsp. flavinogenie W14-3 and its expression in Saccharomyces sp. W0 for Ethanol production. MolBiotechnol57, 337–347.
- Zhang, T., Chi, Z.M., Sheng, J.(2009) A highly thermosensitive and permeable mutant of the marine yeast Cryptococcus aureus G7a potentially useful for single-cell protein production and its nutritive components.Mar Biotechnol12, 280-286.












