Keywords
Breast cancer; Diffuse optical spectroscopy; Optical properties of biological tissue; Clinical trial
Introduction
Breast near-infrared (NIR) diffuse optical tomography (DOT) and diffuse optical spectroscopy (DOS) have been used as novel methods for the characterization and detection of breast tumors [1,2]. In DOS and DOT, the optical properties of the biological tissues, primarily the absorption and the scattering coefficient of the tissue, are measured in the NIR range at different locations of the breast [3-5]. In DOT imaging systems, sources and detectors are placed around the object to image it in different geometric configurations [6]. For image reconstruction, the absorption coefficient at each voxel arises from the summation of the absorption for different chromophores or dyes at each voxel [7,8]. Therefore, by using the absorption coefficient, the concentration of each chromophore at each voxel can be reconstructed. The main concentration constituents in breast tissue that can be reconstructed from the absorption coefficient are oxy-hemoglobin, deoxy-hemoglobin, water, and fat [9-11]. The oxygen saturation in the tissue can be calculated by measuring the concentration of oxy-hemoglobin and deoxyhemoglobin [2,12].
One of the earliest clinical studies involving the use of DOT to detect breast cancer was conducted by Colak et al. [13]. The main purpose of their study was to detect in-homogeneities in breast tissue in vivo using Philips' optical mammography prototype system [14]. The experiment was conducted on 14 subjects, which 4 measurements failed due to experimental malfunction. The measured results showed that the system is capable of detecting in-homogeneities with about 90% success rate [13]. Another group investigated DOT’s ability to monitor the effects of chemotherapy on cancerous lesions [15,16]. Also, There are a number of handheld diffuse optical probes which are implemented and tested to diagnose breast cancer and screening, and reported in [4,17-25].
In this paper, we present the clinical results of a newly developed radio frequency modulated diffuse optical spectroscopy probe. In order to evaluate the performance of the probe, an experimental study was performed on optical phantoms, which mimic the optical properties of the breast tissue. The results of the phantoms study demonstrated that the introduced probe and method accurately measure the optical properties of biological tissue in vivo and that there is no need to use laser diodes or fiber-coupled laser diodes as a light source [23]. The promising results from phantoms study motivated us to test the probe over cancerous tissue in vivo. Therefore, we applied the techniques over malignant breast tumors using contralateral healthy body tissue as a control. This research was conducted in collaboration with the Fraser Health and Jim Pattison Outpatient Care and Surgery Centre-JPOCSC. The clinical study was performed under the Fraser Health Research Ethics Board and Simon Fraser University Ethics Board (SFU study Number: 2015s0156, FHREB Number: FHREB2014-065, principals: Dr. Farid Golnaraghi, Simon Fraser University, and Dr. Rhonda Janzen, JPOCSC).
Materials and Methods
Diffuse optical spectroscopy probe instrumentation
In this study, we have used a newly designed hand-held continuous wave radio frequency modulated diffuse optical spectroscopy (RF-DOS) system to measure optical properties of breast tissue [23]. The RF-DOS instrument and the probe layout are presented in Figures 1a and 1b.
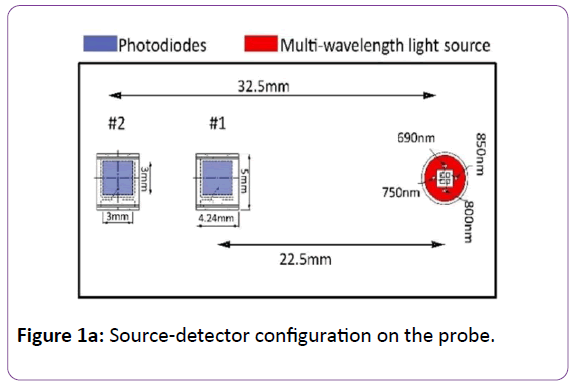
Figure 1a: Source-detector configuration on the probe.

Figure 1b: Photograph of the RF-DOS probe.
In the RF-DOS probe, we used an encapsulated LED light source that emits four wavelengths in the near-infrared range including 690 nm, 750 nm, 800 nm and 850 nm (Marubeni America Corporation L690/750/800/850). These particular wavelengths are selected because breast fat has a minimum absorption coefficient at the mentioned wavelengths [26] which is the main constituent of the breast tissue (57%) [10]. The illuminated light penetrates in the breast tissue and backscattered light is detected via two PIN photodiodes (Vishay Semiconductor Inc. TEMD5010X01) that have high photosensitivity, high radiant sensitivity, and fast response times for near-infrared radiation, thus making them suitable for our application. These two photodiodes are placed 22.5 mm and 32.5 mm away from the light source (eLED).
The intensity of the LEDs with wavelengths of 690 nm, 750 nm, 800 nm and 850 nm are modulated with 8 kHz, 10 KHz, 12 KHz and 14 KHz, respectively. An impedance spectroscope (Zurich Instruments Inc.-HF2IS) is used to generate the required signals for the light sources, collect measured signals, process them, and then pass the results to the host computer. The intensity of light sources is modulated to allow the device to illuminate the breast tissue simultaneously with all wavelengths, and improve the rejection of ambient light and noises. The device design and implementation are fully explained by Shokoufi and Golnaraghi [23].
Protocol
This study was conducted at JPOCSC on fourteen patients. After initial interviews and following the ethical approval protocols, the patients were asked to lie down on their backs, the skin where the probe was to be placed was cleaned with alcohol prep pads prior to the application of the probe, and then, we placed the probe on the location of their tumor. The same procedure was performed on the contralateral side of the breast. Each test procedure took approximately 10 min.
Study population
In this study, a performance of the RF-DOS probes is successfully investigated in an initial clinical trial on subjects who were diagnosed with breast tumor. We collected preliminary data with RF-DOS probes while the probe was located on the breast where diagnosed to have malignant tissue and its contralateral location on the healthy breast. The objective of this study is to process and analyse collected data with the probe and explore performance of the probes to diagnose and characterize cancerous lesion in the breast, in vivo. Table 1 briefly presents the information of patients who were recruited in the study.
| Number of Patients |
Age/Sex |
Tumor Position |
Tumor size |
Tumor type |
| 1 |
53/F |
Left Breast, 3-4O’clock |
2.5*0.8*0.8cm |
BIRADS : 4a |
| 2 |
62/F |
Left Breast, 1O’clock |
2.2*1.7*1.7cm |
BIRADS: 4a |
| 3 |
84/F |
Right Breast, 9O’clock |
10 mm |
Non-invasive |
| 4 |
79/F |
Left Breast, 2O’clock |
7 mm |
BIRADS: 4b |
| 5 |
44/F |
Right Breast, 1O’clock |
2.5*1.7*3.5cm |
BIRADS: 5 |
| 6 |
42/F |
Right Breast, 10-11O’clock |
24 mm |
BIRADS: 4b |
| 7 |
59/F |
Left Breast, 12O’clock |
23*22*15mm |
BI-RADS 4C |
| 8 |
56/F |
Left Breast, 2-3O’clock |
1.6 cm |
BI-RADS: 4a |
| 9 |
63/F |
Left Breast, 5O’clock |
12*9*9 mm |
BI-RADS: 5 |
| 10 |
53/F |
Left Breast, 3-4O’clock |
1.7*1.2*2.4cm |
BI-RADS: 5 |
| 11 |
57/F |
Right Breast, 1o’clock |
12*8*8mm |
BI-RADS : 4c |
| 12 |
54/F |
Right Breast, 12o’clock |
15 mm |
BI-RADS : 5 |
| 13 |
61/F |
Right nipple |
N/A |
Paget’s disease |
| 14 |
78/F |
Left Breast, 3o’clock |
13*11*11mm |
BI-RADS 4B |
Table 1: Brief clinical reports of the patients who have voluntarily enrolled in this study.
Clinical Results
After verification of the developed RF-DOS probe, using phantoms as described by Shokoufi and Golnaraghi [23], the next step was to test the RF-DOS probe on breast tissue, in vivo. To verify performance of the probe on both malignant and healthy breast tissues, we examined fourteen volunteers who have been diagnosed with breast cancer. In order to negate the effects of body movement caused by heartbeat and respiration, the measurement was performed five times. Figure 2 presents the mean and standard deviation of measured intensity and the extracted absorption coefficient for four different wavelengths for patient number 2 while the probe positioned on cancer’s location and its contralateral healthy site. The blue and the yellow bars are the extracted data for left and right breasts, respectively.
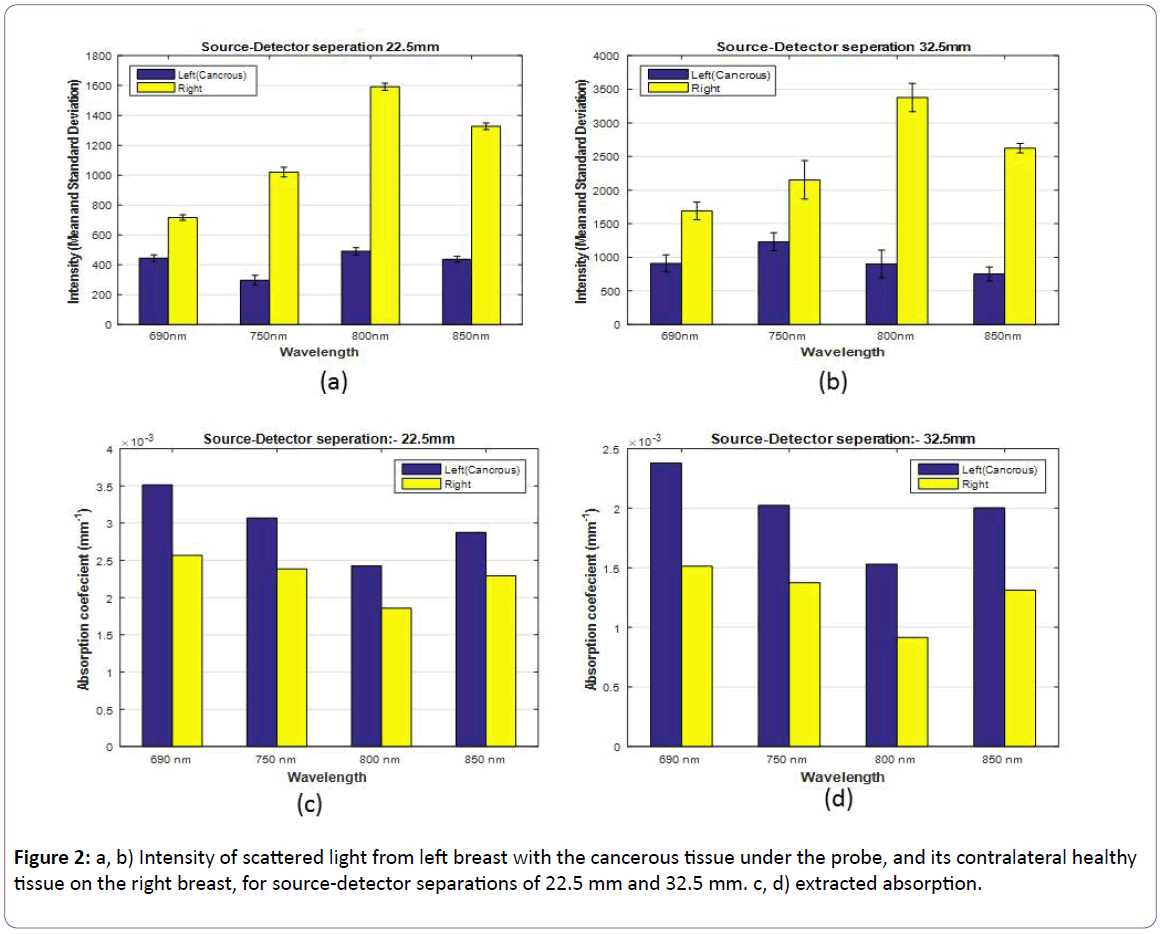
Figure 2: a, b) Intensity of scattered light from left breast with the cancerous tissue under the probe, and its contralateral healthy tissue on the right breast, for source-detector separations of 22.5 mm and 32.5 mm. c, d) extracted absorption.
Patient 2: The NIR data were obtained from a 62 year old female volunteer with breast cancer. According to the clinical data, she was diagnosed with two solid lobulated irregular masses located in the left breast at 3 O’clock and 4 O’clock position. After performing the test, we were able to extract absorption coefficients for both the malignant and healthy breast tissue, using raw data collected from the two detectors located 22.5 mm and 32.5 mm, respectively from the light source (eLED).
The results show that the absorption coefficient of the malignant breast was more than the healthy breast.
Discussion
A total of 28 NIR data acquisitions were performed on 14 tumor locations and their contralateral on healthy breasts. In a study by Shokoufi and Golnaraghi [23], the RF-DOS probe was tested on phantoms made of breast tissue-like material and their ability to measure optical properties was promising. The initial clinical study results are presented in Figure 3 in the form of bar graph. Figures 3a and 4b show the ratio of the backscattered intensity (tumor/healthy) measured at 22.5 mm and 32.5 mm away from the light source, respectively.
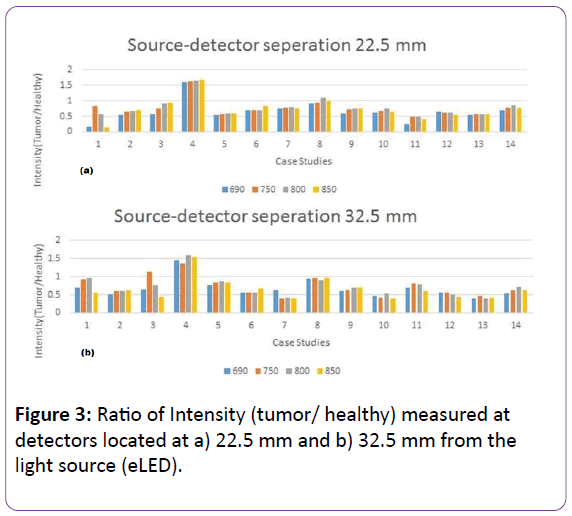
Figure 3: Ratio of Intensity (tumor/ healthy) measured at detectors located at a) 22.5 mm and b) 32.5 mm from the light source (eLED).
Overall, the results show that the breasts with cancer have decreased backscattered light intensity compared to the healthy breasts, for all the four wavelengths (690 nm, 750 nm, 800 nm and 850 nm). Thus, the ratio of intensities, with tumors to without, should be less than one.
A deeper understanding of the physiological differences between malignant and healthy breast tissues is further explored by comparing the absorption coefficients between individual patients. The aspect ratio of the absorption coefficient of the healthy breast to the cancerous one is shown in Figure 4.
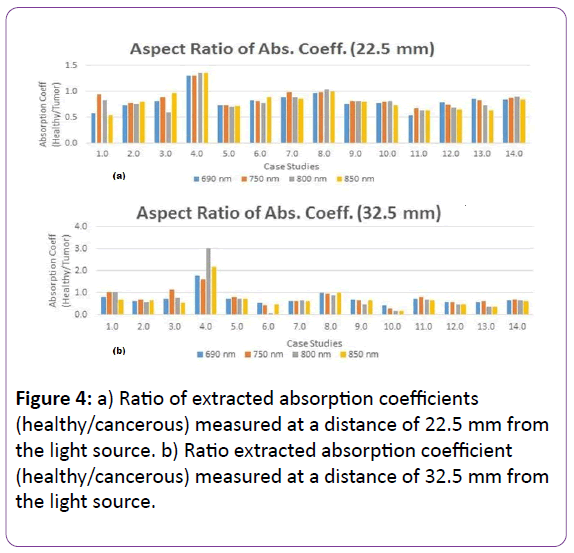
Figure 4: a) Ratio of extracted absorption coefficients (healthy/cancerous) measured at a distance of 22.5 mm from the light source. b) Ratio extracted absorption coefficient (healthy/cancerous) measured at a distance of 32.5 mm from the light source.
Figures 4a and 5b also show that the ratio of the absorption coefficients for the breast with malignancy at four wavelengths (690 nm, 750 nm, 800 nm and 850 nm) in both locations (22.5 mm and 32.5. mm) are higher than normal tissue, due to their increased vascularization, which is consistent with the literature, except for case study four. The probe did not detect the tumor due to its small size (7 mm in diameter) and depth, located nine cm away from the nipple. The results from the comparison bar graphs of absorption coefficients for all the patients show a promising detection rate (sensitivity) of 92 percent while the mammography sensitivity is around 67 present [2].
Concentration of Chromophores
Four main chromophores contribute to the attenuation at wavelength λ in breast tissue, and the multi-spectral directapproach method is employed to extract the two main absorbers’ concentration in two different path lengths for r=22.5 mm and r=32.5 mm [27].
Figure 5 shows Hb and HbO2 concertation ratio for healthy and diseased tissue for two path lengths.
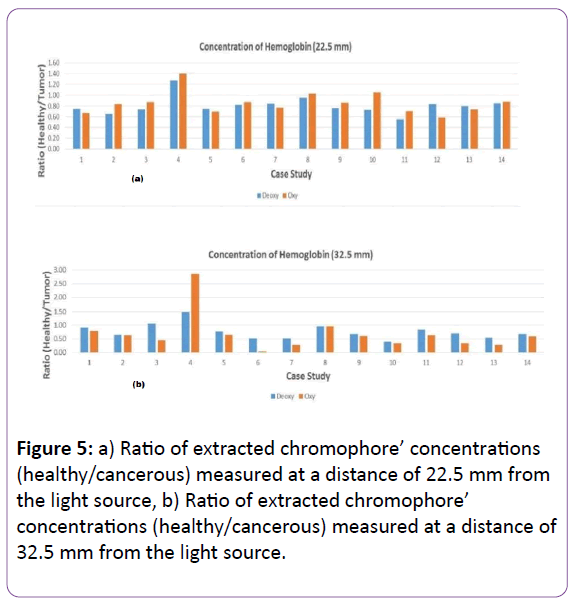
Figure 5: a) Ratio of extracted chromophore’ concentrations (healthy/cancerous) measured at a distance of 22.5 mm from the light source, b) Ratio of extracted chromophore’ concentrations (healthy/cancerous) measured at a distance of 32.5 mm from the light source.
As Figure 5 shows, these parameters vary in cancerous and healthy tissue due to a higher level of absorption caused by greater vascularization in cancerous tissue. For all patients, the average value of oxy-hemoglobin and deoxy-hemoglobin concentrations (healthy/cancerous) is less than one, which shows the higher concentration of these two main chromophores in the cancerous tissue except case study number four, as previously explained.
Table 2 summarizes the clinical trial results of fourteen patients using RF-DOS probe.
| Case study |
Cancerous breast |
Description |
| 1 |
Left |
Tumor correctly detected |
| 2 |
Left |
Tumor correctly detected |
| 3 |
Left |
Tumor correctly detected |
| 4 |
Right |
Tumor is not detected due to depth of tumor |
| 5 |
Left |
Tumor correctly detected |
| 6 |
Right |
Tumor correctly detected |
| 7 |
Right |
Tumor correctly detected |
| 8 |
Left |
Tumor correctly detected |
| 9 |
Left |
Tumor correctly detected |
| 10 |
Left |
Tumor correctly detected |
| 11 |
Right |
Tumor correctly detected |
| 12 |
Right |
Tumorcorrectly detected |
| 13 |
Right |
Tumor correctly detected |
| 14 |
Left |
Tumor correctly detected |
Table 2: Summary of clinical trial results using DOS probe.
The developed diffuse optical spectroscopy probe is capable of providing the optical data (absorption and scattering coefficients) of tissue, as well as their changes in the breast tissue with the help of developed software and hardware. As the results were consistent with the literature, this study conclusively proves that the RF-DOS probe can differentiate between malignant and healthy breast tissue in vivo.
For these experiments, we used off self-hardware for data acquisition and signal generation which is heavy and bulky. In the next phase of the project, we will be redesigning the device to be more compact, with all the components integrated into a single device, which will be more ergonomic and user-friendly. The intensity of the light source will be variable and further development will be done to automate the device to reduce operational error.
Currently, data analysis and coefficient extraction from the raw data are performed separately In order to construct a real time device, we will add data analysis and coefficient extraction algorithms to our software to have real time data visualization.
Conclusion
In our previous study by Shokoufi and Golnaraghi [23], we had tested the probe on tissue-like materials and the results were promising regarding its ability to measure their optical properties. In this paper, we reported clinical results obtained with recently developed radio frequency modulated diffuse optical spectroscopy (DOS-RF) probe to measure optical properties of breast tissue, in vivo. Fourteen patients who diagnosed with breast cancer conducted in this study voluntarily. The optical properties of cancerous tissue and its contralateral healthy site were measured, in order to have comparison between them.
The results showed that in 92 percent of cases, absorption coefficient of cancerous tissue is higher than healthy tissue at almost all the wavelengths (690 nm, 750 nm, 800 nm and 850 nm). The extracted data demonstrated that RF-DOS probe can identify the cancerous region from the surrounding healthy tissue, as the cancerous regions have higher absorption coefficient due to higher levels of vascularization in comparison to healthy tissue. The clinical results have correlated well with the cancer locations obtained by other clinical modalities such as X-ray mammography and MRI. We have to perform several clinical studies on different subject population (healthy, benign and suspicions lesion) to calculate accuracy, specificity and positive predictive value for the probe.
Acknowledgement
Financial support for this project was provided in-part by the Natural Sciences and Engineering Research Council of Canada (NSERC). The authors thank Dr. Rhonda Janzen (from Jim Pattison Outpatient Care and Surgery Centre) and Dr. Mojgan Jenab (from SFU) for their assistance and support of this study.
21315
References
- Hassan A, El-shenawee M (2011) Review of electromagnetic techniques for breast cancer detection. IEEE Rev Biomed Eng 4:103-118.
- Foschum F, Kienle A (2012) Broadband absorption spectroscopy of turbid media using a dual step steady-state method. J Biomed Opt 17: 37009.
- Cerussi A, Shah N, Hsiang D, Durkin A, Butler J, et al. (2012) In vivoabsorption, scattering and physiologic properties of 58 malignant breast tumors determined by broadband diffuse optical spectroscopy. J Biomed Opt 11: 44005.
- Boas DA, Pitris C, Ramanujam N (2011) Handbook of Biomedical Optics. CRC Press.
- Konecky SD (2008) Non-invasive imaging of breast cancer with diffusing near-infrared light.
- Farrell TJ (1992) A diffusion theory model of spatially resolved, steady-state diffuse reflectance for the non-invasive determination of tissue optical properties in vivo. Med Phys 19:879.
- Jacques SL, Pogue BW (2013) Tutorial on diffuse light transport. J Biomed Opt 13:41302.
- Troy TL, Page DL, Sevick-muraca EM (1996) Optical properties of normal and diseased breast t issues: Prognosis for optical mammography. J Biomed Opt 1: 342-355.
- Durduran T, Choe R, Culver JP, Zubkov L, Holboke MJ, et al. (2002) Bulk optical properties of healthy female breast tissue. Phys Med Biol 47: 2847-2286.
- Gibson AP, Hebden JC, Arridge SR (2014) Recent advances in diffuse optical imaging. Phys Med Biol 50: R1-43.
- Colak SB, van der Mark MB, Hooft GW, Hoogenraad JH, van der Linden ES, et al. (1999) Clinical optical tomography and NIR spectroscopy for breast cancer detection. IEEE J Sel Top Quantum Electron 5: 1143-1158.
- Hoogenraad JH, van der Mark MB, Colak S-BB,T’Hooft GW, van der Linden ES (1997) First results from the Philips optical mammoscope. SPIE, ProcPhotPropag Tissues III 3194: 184-190.
- Soliman H, Gunasekara A, Rycroft M, Zubovits J, Dent R, et al. (2010) Functional imaging using diffuse optical spectroscopy of neoadjuvant chemotherapy response in women with locally advanced breast cancer. Clin Cancer Res 16: 2605-2614.
- Tromberg BJ, Cerussi AE (2010) Imaging breast cancer chemotherapy response with light: Commentary on soliman.ClinCancer Res 16:2486-2488.
- Flexman ML, Kim HK, Stoll R, Khalil M, Fong CJ, et al. (2012) A wireless handheld probe with spectrally constrained evolution strategies for diffuse optical imaging of tissue. Rev Sci83: 33108.
- Biswas SK, Rajan K, Vasu RM (2009) Diffuse optical tomographic imager using a single light source. J ApplPhys 105:24702.
- Erickson SJ, Godavarty A, Martinez SL, Gonzalez J, Romero A, et al. (2012) Hand-held optical devices for breast cancer: Spectroscopy and 3-D tomographic imaging. IEEE J Sel Top Quantum Electron 18: 1298-1312.
- Erickson SJ, Godavarty A (2009) Hand-held based near-infrared optical imaging devices: a review. Med EngPhys 31: 495-509.
- No KS, Xie Q, Kwong R, Cerussi A, Tromberg BJ, et al. (2006) HBS: A handheld breast cancer detector based on frequency domain photon migration with full heterodyne.IEEE Biomed Circuits SystConf114-117.
- Yang H, Xi L, Samuelson S, Xie H, Yang L, et al. (2013) Handheld miniature probe integrating diffuse optical tomography with photoacoustic imaging through a MEMS scanning mirror. Biomed Opt Express 4:427-432.
- Shokoufi M, Golnaraghi F (2015) Development of a handheld diffuse optical breast cancer assessment probe. J Innov Opt Health Sci 9: 1-10.
- Godavarty A, Rodriguez S, Jung Y-J, Gonzalez S (2015) Optical imaging for breast cancer pre-screening. Breast Cancer Targets Ther 2015: 193-209.
- Zhu Q, Tannenbaum S, Kurtzman S. (2007) Optical tomography with ultrasound localization for breast cancer diagnosis and treatment monitoring. SurgOncolClin N Am 16: 307-321.
- Flexman M (2012) Dynamic digital optical tomography for cancer imaging and therapy monitoring. Vasa. Columbia University.
- Shokoufi M (2016) Multi-modality breast cancer assessment tools using diffuse optical and electrical impedance spectroscopy. Simon Fraser University.











